
API's
Active Pharmaceutical Ingredients

Active Pharmaceutical Ingredients
API
BP / USP / EP
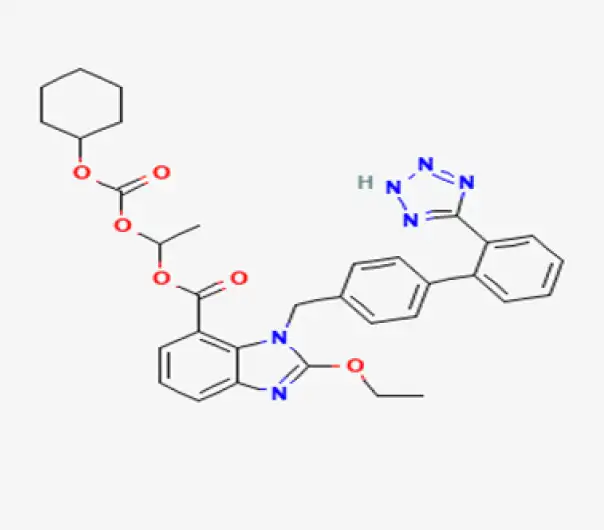
C26H28N6O6
Ethyl 5-(cyclohexyloxycarbonyloxy)-1-[[2'-(1H-tetrazol-5-yl)biphenyl-4-yl]methyl]-2,3-dihydro-1H-indole-2-carboxylate
145040-37-5
440.52 g/mol
Angiotensin II receptor blocker (ARB) prodrug
Antihypertensive / Cardiovascular agent
| Appearance | White to off-white crystalline powder |
|---|---|
| Solubility | Freely soluble in chloroform and acetone; slightly soluble in ethanol; practically insoluble in water |
| Melting Point | 220–230°C |
| pH (1% aqueous solution) | - |
Candesartan cilexetil is an angiotensin II receptor blocker (ARB) prodrug used for hypertension and heart failure. After oral administration it is rapidly converted to candesartan, providing long-duration blood pressure control with excellent tolerability and a low incidence of cough or angioedema—useful for patients intolerant to ACE inhibitors.
Candesartan Cilexetil works by selectively blocking the AT₁ receptors of angiotensin II, a potent vasoconstrictor: Vasodilation: Inhibits angiotensin II–mediated blood vessel constriction, Reduced aldosterone secretion: Lowers sodium and water retention
| Attribute | Why Salius Pharma Is a Good Choice |
|---|---|
| Quality Certification | WHO GMP, ISO 9001:2015, FDA audited facilities |
| Affordability & Generic Access | Cost competitive generics for global supply |
| Export Experience | Proven track record of supplying globally |
When you order from Salius Pharma, you receive more than just a product — you receive a complete, professionally prepared package that meets international regulatory and quality expectations.
We ensure prompt and reliable shipping worldwide. Delivery times vary depending on destination, order volume, and regulatory requirements.
7 to 21 business days from dispatch, depending on your location.
Store in airtight containers, protected from moisture and light, at controlled room temperature.
White to off-white crystalline powder; stable under normal conditions; moisture-sensitive—control humidity during handling.
Yes. Common combinations include Hydrochlorothiazide for improved blood pressure control.
COA (per BP/USP/EP), MSDS, TSE/BSE statements, Stability Data, GMP certificates, and DMF/CTD dossiers as applicable.
Yes. It is widely used as a long-term maintenance therapy for hypertension and heart failure with strong evidence supporting its safety and efficacy.
Looking to source Candesartan Cilexetil or other high-quality pharmaceutical products?
We’re here to help.
Whether you need Active Pharmaceutical Ingredients (APIs), finished formulations, or regulatory support,
our team is ready to provide dependable, compliant, and cost-effective solutions tailored to your market needs.
The contents of this page and any attachments are intended solely for the designated recipient(s) and may contain confidential and/or privileged information protected by law.
Any patented products are excluded from our offerings in regions where such patents are currently in force.