
API's
Active Pharmaceutical Ingredients

Active Pharmaceutical Ingredients
API
BP/USP
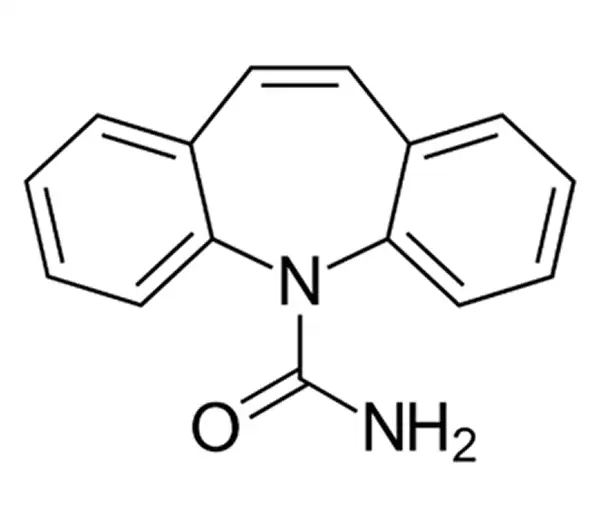
C15H12N2O
2-azatricyclo[9.4.0.0^{3,8}]pentadeca-1(15),3,5,7,9,11,13-heptaene-2-carboxamide
298-46-4
236.27 g/mol
Dibenzazepines
Miscellaneous Anticonvulsants
| Appearance | White or almost white crystalline powder |
|---|---|
| Solubility | Practically insoluble in water; soluble in acetone, chloroform, and ethanol; slightly soluble in methanol |
| Melting Point | 189–193 °C |
| pH (1% aqueous solution) | 5.0–6.0 |
Carbamazepine, also known as Tegretol, is an anticonvulsant and analgesic drug used to control seizures and to treat trigeminal neuralgia pain. It is also prescribed to manage bipolar disorder, making it one of the first anticonvulsants to be used as a mood stabilizer. Approved by the FDA in 1965, Carbamazepine remains a cornerstone therapy for epilepsy and neuropathic pain management.

Carbamazepine acts primarily by inhibiting voltage-gated sodium channels in the brain, stabilizing hyperexcited neuronal membranes and reducing repetitive neuronal firing. This decreases synaptic transmission, thereby preventing seizure propagation and stabilizing mood in bipolar disorder.
| Attribute | Why Salius Pharma Is a Good Choice |
|---|---|
| Quality Certification | WHO GMP, ISO 9001:2015, FDA audited facilities |
| Affordability & Generic Access | Cost competitive generics for global supply |
| Export Experience | Proven record of supplying globally |
When you order from Salius Pharma, you receive more than just a product — you receive a complete, professionally prepared package that meets international regulatory and quality expectations.
We ensure prompt and reliable shipping worldwide. Delivery times vary depending on destination, order volume, and regulatory requirements.
7 to 21 business days from dispatch, depending on your location.
Salius Pharma supplies Carbamazepine Anhydrous API (≥99% purity), compliant with BP/USP standards, to markets including Mexico, Argentina, Colombia, Peru, Bolivia, and Guatemala under WHO-GMP certification.
Salius offers DMFs, CoAs, and CTD/eCTD dossiers tailored for regulatory agencies across global markets, including Latin America and the Middle East.
Yes. It is commonly prescribed for chronic conditions such as epilepsy and bipolar disorder. However, periodic monitoring for side effects and dose adjustments are necessary.
It blocks sodium channels in neuronal membranes, reducing excessive electrical activity in the brain, helping control seizures, nerve pain, and mood stabilization.
Common side effects include drowsiness, nausea, dizziness, and unsteadiness. Rare but serious effects include skin reactions (SJS/TEN), blood disorders, and drug interactions.
Looking to source Carbamazepine or other high-quality pharmaceutical products?
We’re here to help.
Whether you need Active Pharmaceutical Ingredients (APIs), finished formulations, or regulatory support,
our team is ready to provide dependable, compliant, and cost-effective solutions tailored to your market needs.
The contents of this page and any attachments are intended solely for the designated recipient(s) and may contain confidential and/or privileged information protected by law.
Any patented products are excluded from our offerings in regions where such patents are currently in force.