
API's
Active Pharmaceutical Ingredients

Active Pharmaceutical Ingredients
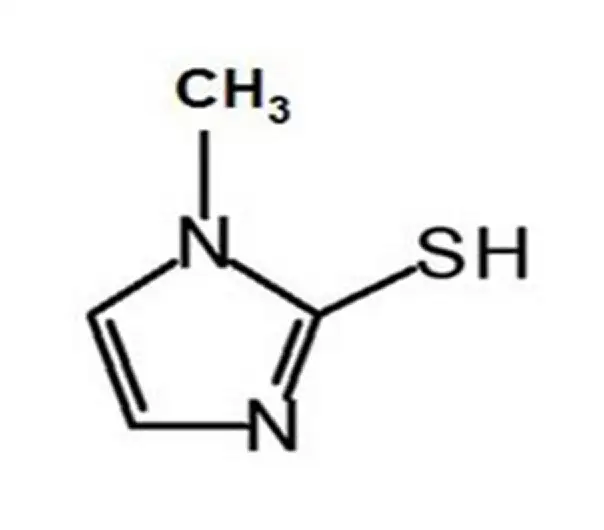
API
USP / EP / BP (as required)
C4H6N2S
1-methyl-1H-imidazole-2-thiol
60-56-0
114.17 g/mol
Thionamide – Antithyroid Agent
| appearance | White to off-white crystalline powder |
|---|---|
| solubility | Freely soluble in water; Soluble in alcohol; Slightly soluble in ether and chloroform |
| melting_point | 146–150°C |
| ph | ≈5.0 – 6.5 |
Methimazole is a potent antithyroid API used to manage hyperthyroidism (including Graves\u2019 disease and toxic goiter) by inhibiting the synthesis of thyroid hormones T3 and T4.
Inhibits thyroid peroxidase–mediated iodine oxidation, iodination of tyrosine residues, and coupling reactions, thereby reducing thyroid hormone (T3/T4) production and controlling hyperthyroid symptoms.
| Attribute | Why Salius Pharma Is a Good Choice |
|---|---|
| Quality Certification | WHO GMP, ISO 9001:2015, FDA audited facilities |
| Affordability & Generic Access | Cost competitive generics for global supply |
| Export Experience | Proven track record of supplying globally |
When you order from Salius Pharma, you receive more than just a product — you receive a complete, professionally prepared package that meets international regulatory and quality expectations.
We ensure prompt and reliable shipping worldwide. Delivery times vary depending on destination, order volume, and regulatory requirements.
7 to 21 business days from dispatch, depending on your location.
WHO-GMP manufacturing, ISO-certified quality systems, FDA-audited facilities, full documentation support, and complete batch traceability for regulated and semi-regulated markets.
Yes. Salius supports submissions for stringent markets with the required regulatory files.
Yes. Potential interactions include anticoagulants, beta-blockers, and digoxin. Appropriate warnings should be included per regulatory guidance.
Asia, Africa, Middle East, Latin America, CIS, and several regulated markets, with support for local regulatory submissions.
Competitive pricing, flexible MOQs, fast export timelines, strong logistics, end-to-end documentation, and certified facility quality.
7–14 business days for in-stock material; 14–21 days when additional documentation/inspection is needed. Transit time varies by destination and shipping mode.
Looking to source Methimazole or other high-quality pharmaceutical products?
We’re here to help.
Whether you need Active Pharmaceutical Ingredients (APIs), finished formulations, or regulatory support,
our team is ready to provide dependable, compliant, and cost-effective solutions tailored to your market needs.
The contents of this page and any attachments are intended solely for the designated recipient(s) and may contain confidential and/or privileged information protected by law.
Any patented products are excluded from our offerings in regions where such patents are currently in force.