
API's
Active Pharmaceutical Ingredients

Active Pharmaceutical Ingredients
API
IP / BP / USP / EP
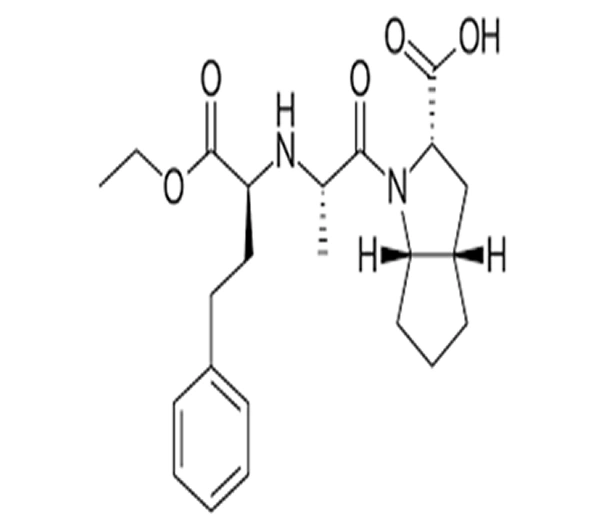
C23H32N2O5
(2S,3aS,6aS)-1-[(2S)-2-[[(2S)-1-ethoxy-1-oxo-4-phenylbutan-2-yl]amino]propanoyl]octahydrocyclopenta[b]pyrrole-2-carboxylic acid
87333-19-5
416.52 g/mol
ACE Inhibitor (Antihypertensive)
| Appearance | White to off-white crystalline powder |
|---|---|
| Solubility | Slightly soluble in water; freely soluble in methanol and ethanol |
| Melting Point | 105–112°C |
Ramipril is a widely used antihypertensive medication belonging to the ACE inhibitor class. It is primarily prescribed for the treatment of hypertension, heart failure, diabetic nephropathy, and cardiovascular risk reduction. Ramipril is a prodrug that is rapidly converted to its active metabolite Ramiprilat, which exhibits strong ACE-inhibitory activity.
Ramipril inhibits the angiotensin-converting enzyme (ACE), blocking the conversion of angiotensin I to angiotensin II. This results in vasodilation, reduced blood pressure, decreased preload and afterload, and lower aldosterone secretion. These effects make it highly effective for hypertension, heart failure management, and cardiovascular protection.
| Attribute | Why Salius Pharma Is a Good Choice |
|---|---|
| Quality Certification | WHO GMP, ISO 9001:2015, FDA audited facilities |
| Affordability & Generic Access | Cost competitive generics for global supply |
| Export Experience | Proven track record of supplying globally |
When you order from Salius Pharma, you receive more than just a product — you receive a complete, professionally prepared package that meets international regulatory and quality expectations.
We ensure prompt and reliable shipping worldwide. Delivery times vary depending on destination, order volume, and regulatory requirements.
7 to 21 business days from dispatch, depending on your location.
We export globally — Asia, Europe, Africa, CIS, Middle East, and South America.
COO, GMP, FSC (where applicable), ISO certificates, and other required paperwork.
Orders can be placed via email, WhatsApp, or company purchase order system.
Yes. Annual contracts, long-term volume agreements, and supply partnerships are available.
We ship via air freight, sea freight based on the customer’s urgency, order size, and destination regulations.
Delivery is usually completed in 7–21 business days depending on stock availability, documentation, and destination
Looking to source Ramipril or other high-quality pharmaceutical products?
We’re here to help.
Whether you need Active Pharmaceutical Ingredients (APIs), finished formulations, or regulatory support,
our team is ready to provide dependable, compliant, and cost-effective solutions tailored to your market needs.
The contents of this page and any attachments are intended solely for the designated recipient(s) and may contain confidential and/or privileged information protected by law.
Any patented products are excluded from our offerings in regions where such patents are currently in force.