
API's
Active Pharmaceutical Ingredients

Active Pharmaceutical Ingredients
API
IP / BP / USP / EP
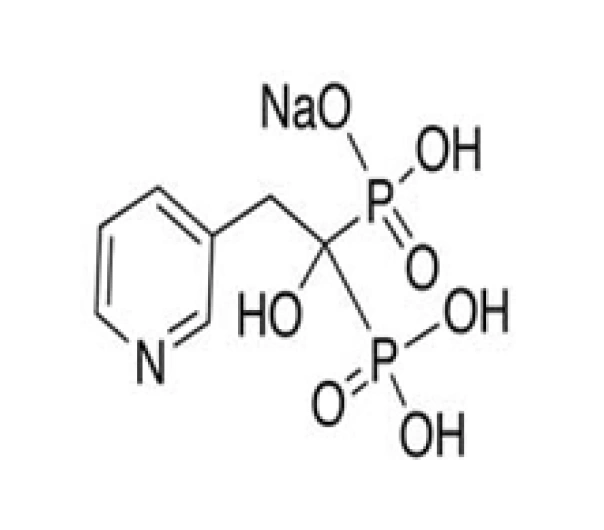
C7H10NO7P2 • Na
Sodium 4-[(1-hydroxy-1-phosphono-2-pyridinyl)methyl]phosphonate
115436-72-1
305.10 g/mol
Bisphosphonate (Bone Resorption Inhibitor)
| Appearance | White to off-white powder |
|---|---|
| Solubility | Freely soluble in water; Practically insoluble in organic solvents |
| Melting Point | 250°C (decomposes) |
| pH (1% solution) | 3.5–5.0 (1% aqueous solution) |
Risedronate Sodium is a potent bisphosphonate used primarily for the treatment and prevention of osteoporosis. It works by inhibiting osteoclast-mediated bone resorption, thereby increasing bone mineral density. It is widely used in formulations for post-menopausal osteoporosis, glucocorticoid-induced osteoporosis, and Paget’s disease. Its favorable safety and efficacy profile makes it a commonly preferred therapeutic option.
Risedronate binds strongly to hydroxyapatite crystals in bone and inhibits osteoclast activity. This leads to reduced bone turnover, increased bone density, and lower fracture risk. It has high affinity for bone tissue and a long duration of action.
| Attribute | Why Salius Pharma Is a Good Choice |
|---|---|
| Quality Certification | WHO GMP, ISO 9001:2015, FDA audited facilities |
| Affordability & Generic Access | Cost competitive generics for global supply |
| Export Experience | Proven track record of supplying globally |
When you order from Salius Pharma, you receive more than just a product — you receive a complete, professionally prepared package that meets international regulatory and quality expectations.
We ensure prompt and reliable shipping worldwide. Delivery times vary depending on destination, order volume, and regulatory requirements.
7 to 21 business days from dispatch, depending on your location.
It is used for treating and preventing osteoporosis and Paget’s disease.
COO, GMP, FSC (where applicable), ISO certificates, and other required paperwork.
Orders can be placed via email, WhatsApp, or company purchase order system.
Yes. Annual contracts, long-term volume agreements, and supply partnerships are available.
We ship via air freight and sea freight based on the customer’s urgency, order size, and destination regulations.
Delivery is usually completed in 7–21 business days depending on stock availability, documentation, and destination
Looking to source Risedronate Sodium or other high-quality pharmaceutical products?
We’re here to help.
Whether you need Active Pharmaceutical Ingredients (APIs), finished formulations, or regulatory support,
our team is ready to provide dependable, compliant, and cost-effective solutions tailored to your market needs.
The contents of this page and any attachments are intended solely for the designated recipient(s) and may contain confidential and/or privileged information protected by law.
Any patented products are excluded from our offerings in regions where such patents are currently in force.