
API's
Active Pharmaceutical Ingredients

Active Pharmaceutical Ingredients
API
BP/USP
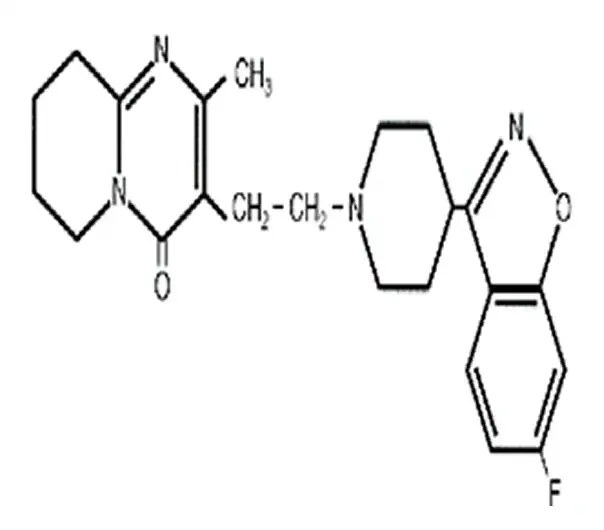
C₂₃H₂₇FN₄O₂
[3-[2-[4-(6-fluoro-1,2-benzisoxazol-3-yl)piperidin-1-yl]ethyl]-2-methyl-6,7,8,9-tetrahydro-4H-pyrido[1,2-a]pyrimidin-4-one]
106266-06-2
410.49 g/mol
Atypical (second-generation) antipsychotics
| Appearance | White to off-white crystalline powder |
|---|---|
| Solubility | Poorly soluble in water; soluble in organic solvents like methanol, ethanol, and chloroform |
| Melting point | 170–175°C |
| pH | 5–9 |
Risperidone is an atypical antipsychotic medication used to treat schizophrenia, bipolar disorder, and irritability associated with autism. It helps control mood, thought disturbances, and behavioral symptoms.
Risperidone works by balancing neurotransmitters in the brain, primarily blocking dopamine (D2) and serotonin (5-HT2) receptors. This dual mechanism helps reduce psychotic symptoms while improving mood stability.
| Attribute | Why Salius Pharma Is a Good Choice |
|---|---|
| Quality Certification | WHO GMP, ISO 9001:2015, FDA audited facilities |
| Pharmacopoeial Compliance | Meets BP/USP standards |
| Therapeutic Category | Atypical antipsychotic |
When you order from Salius Pharma, you receive more than just a product — you receive a complete, professionally prepared package that meets international regulatory and quality expectations.
We ensure prompt and reliable shipping worldwide. Delivery times vary depending on destination, order volume, and regulatory requirements.
7 to 21 business days from dispatch, depending on your location.
Yes, weight gain is common due to metabolic changes and increased appetite. Monitoring diet and exercise is recommended during treatment.
No, Risperidone is not addictive. However, it should not be stopped abruptly without medical advice to avoid relapse or withdrawal effects.
Some improvements may be seen within a week, but full therapeutic effects typically develop over 4–6 weeks.
Salius Pharma supplies Risperidone as an Active Pharmaceutical Ingredient (API).
To order, contact Salius Pharma with details like required quantity, intended market, and regulatory documentation needs (DMF, GMP certificates, etc.).
Salius Pharma exports Risperidone worldwide, including Asia, Europe, Africa, the Middle East, North America, and South America.
Looking to source Risperidone or other high-quality pharmaceutical products?
We’re here to help.
Whether you need Active Pharmaceutical Ingredients (APIs), finished formulations, or regulatory support,
our team is ready to provide dependable, compliant, and cost-effective solutions tailored to your market needs.
The contents of this page and any attachments are intended solely for the designated recipient(s) and may contain confidential and/or privileged information protected by law.
Any patented products are excluded from our offerings in regions where such patents are currently in force.