
API's
Active Pharmaceutical Ingredients

Active Pharmaceutical Ingredients
API
IP / BP / USP / EP
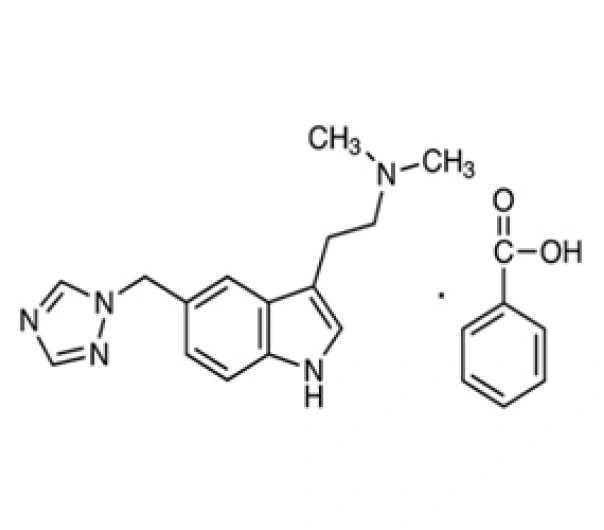
C15H19N5• C7H6O2 (benzoate)
N,N-dimethyl-5-(1H-1,2,4-triazol-1-ylmethyl)-1H-indole-3-ethanamine benzoate
145202-66-0
391.48 g/mol (benzoate salt)
Selective 5-HT1B/1D Receptor Agonist (Anti-migraine Agent)
| Appearance | White to off-white crystalline powder |
|---|---|
| Solubility | Freely soluble in water; soluble in methanol and ethanol; slightly soluble in organic solvents |
| Melting Point | 180–190°C (approx.) |
| pH (1% solution) | Stable in slightly acidic to neutral conditions |
Rizatriptan Benzoate is a fast-acting antimigraine API belonging to the triptan class. It is widely used for acute migraine attacks, with or without aura. The benzoate form enhances stability and solubility, making it suitable for tablet and orally disintegrating tablet (ODT) formulations.
Rizatriptan selectively stimulates 5-HT1B and 5-HT1D receptors, causing cranial blood vessel constriction, inhibition of neuropeptide release, and reduced trigeminal nerve pain signaling. These combined actions provide fast relief from migraine symptoms.
| Attribute | Why Salius Pharma Is a Good Choice |
|---|---|
| Quality Certification | WHO GMP, ISO 9001:2015, FDA audited facilities |
| Affordability & Generic Access | Cost competitive generics for global supply |
| Export Experience | Proven track record of supplying globally |
When you order from Salius Pharma, you receive more than just a product — you receive a complete, professionally prepared package that meets international regulatory and quality expectations.
We ensure prompt and reliable shipping worldwide. Delivery times vary depending on destination, order volume, and regulatory requirements.
7 to 21 business days from dispatch, depending on your location.
The formula is C22H25N5O4.
It is a triptan, specifically a serotonin (5-HT) receptor agonist.
It treats acute migraine attacks with or without aura in adults and adolescents.
It binds to 5-HT1B/1D receptors, causing cranial vasoconstriction and inhibiting pro-inflammatory neuropeptide release.
Freely soluble in methanol, soluble in water and ethanol.
High-quality generics at competitive prices for global markets.
Looking to source Rizatriptan Benzoate or other high-quality pharmaceutical products?
We’re here to help.
Whether you need Active Pharmaceutical Ingredients (APIs), finished formulations, or regulatory support,
our team is ready to provide dependable, compliant, and cost-effective solutions tailored to your market needs.
The contents of this page and any attachments are intended solely for the designated recipient(s) and may contain confidential and/or privileged information protected by law.
Any patented products are excluded from our offerings in regions where such patents are currently in force.