
API's
Active Pharmaceutical Ingredients

Active Pharmaceutical Ingredients
API
IP / BP / USP / EP
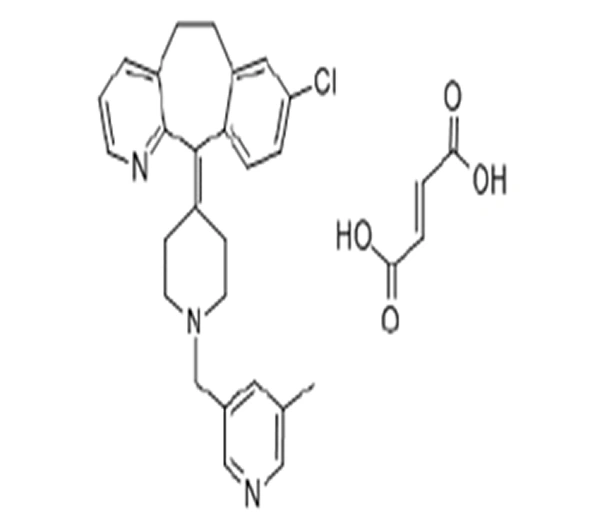
C26H26ClN3O • C4H4O4 (fumarate)
8-chloro-6,11-dihydro-11-[1-[(5-methyl-3-pyridinyl)methyl]-4-piperidinylidene]-5H-benzo[5,6]cyclohepta[1,2-b]pyridine fumarate
158876-82-5
572.10 g/mol
Second-Generation Antihistamine / PAF Antagonist
| Appearance | White to off-white crystalline powder |
|---|---|
| Solubility | Freely soluble in methanol and ethanol; slightly soluble in water; soluble in acidic aqueous media |
| Melting Point | 195–205°C (approx., fumarate form) |
| pH (1% solution) | Stable in slightly acidic to neutral pH |
Rupatadine Fumarate is a non-sedative; second-generation antihistamine used for the treatment of allergic rhinitis and chronic urticaria. It exhibits dual pharmacological action as both a H1-antihistamine and a Platelet-Activating Factor (PAF) antagonist, providing broad anti-allergic activity.
Rupatadine exerts anti-allergic effects through two mechanisms: H1-receptor antagonism, reducing histamine-mediated symptoms such as sneezing, itching, and rhinorrhea; and PAF antagonism, preventing inflammatory cell activation and improving control in chronic allergies. This dual activity makes it highly effective for both seasonal and perennial allergic conditions.
| Attribute | Why Salius Pharma Is a Good Choice |
|---|---|
| Quality Certification | WHO GMP, ISO 9001:2015, FDA audited facilities |
| Affordability & Generic Access | Cost competitive generics for global supply |
| Export Experience | Proven track record of supplying globally |
When you order from Salius Pharma, you receive more than just a product — you receive a complete, professionally prepared package that meets international regulatory and quality expectations.
We ensure prompt and reliable shipping worldwide. Delivery times vary depending on destination, order volume, and regulatory requirements.
7 to 21 business days from dispatch, depending on your location.
It Blocks H1 receptors and PAF to reduce allergic responses.
In-stock orders ship within 3-5 business days.
7-21 business days globally from dispatch.
Asia, Europe, Africa, CIS, Middle East, South America.
WHO-GMP, ISO 9001:2015, FDA-audited facilities.
Star export house status ensures reliability, affordability, and compliance.
Looking to source Rupatadine Fumarate or other high-quality pharmaceutical products?
We’re here to help.
Whether you need Active Pharmaceutical Ingredients (APIs), finished formulations, or regulatory support,
our team is ready to provide dependable, compliant, and cost-effective solutions tailored to your market needs.
The contents of this page and any attachments are intended solely for the designated recipient(s) and may contain confidential and/or privileged information protected by law.
Any patented products are excluded from our offerings in regions where such patents are currently in force.