
API's
Active Pharmaceutical Ingredients

Active Pharmaceutical Ingredients
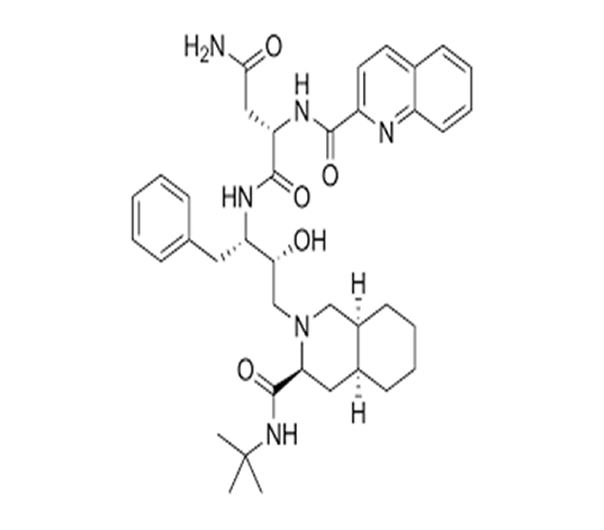
API (Antiretroviral – HIV Protease Inhibitor)
USP / EP / In-house Pharmaceutical Grade
C₃₈H₅₀N₆O₅
(2S)-N-[(2S,3R)-4-[(3S)-3-(tert-butylcarbamoyl)amino-2-hydroxy-4-phenylbutyl]-3-hydroxy-1,3-dioxobutan-2-yl]-2-(quinolin-2-ylformamido)hexanediamide
127779-20-8
670.83 g/mol
HIV-1 Protease Inhibitor (PI Class Antiretroviral)
| Appearance | White to off-white crystalline powder |
|---|---|
| Solubility | Very low water solubility; Soluble in ethanol, methanol, DMSO; Solubility enhanced in acidic environments (salt form) |
| Melting Point | ~210–215°C (Saquinavir Free Base) |
| pKa | ~7.4 (weakly basic compound) |
Saquinavir is a potent HIV-1 protease inhibitor (PI) used in combination antiretroviral therapy (ART). It is typically formulated as Saquinavir Mesylate, a more stable and bioavailable salt form. It is an essential second-line ART drug in many HIV treatment programs worldwide.
Saquinavir inhibits the HIV protease enzyme responsible for: Cleaving viral polyproteins; Maturing viral particles; Enabling viral infectivity. By blocking protease, Saquinavir prevents the formation of mature, infectious HIV particles, reducing viral load significantly. Saquinavir is typically co-administered with Ritonavir to boost bioavailability through CYP3A4 inhibition.
| Attribute | Why Salius Pharma Is a Good Choice |
|---|---|
| Quality Certification | WHO GMP, ISO 9001:2015, FDA audited facilities |
| Affordability & Generic Access | Cost competitive generics for global supply |
| Export Experience | Proven track record of supplying globally |
When you order from Salius Pharma, you receive more than just a product — you receive a complete, professionally prepared package that meets international regulatory and quality expectations.
We ensure prompt and reliable shipping worldwide. Delivery times vary depending on destination, order volume, and regulatory requirements.
7 to 21 business days from dispatch, depending on your location.
Saquinavir is a first-generation HIV-1 Protease Inhibitor (PI) used primarily in combination regimens to suppress viral replication.
Generally, 36–48 months, depending on storage and salt form.
Yes. Strong interactions occur with medications affecting CYP3A4, including antifungals, antibiotics, statins, and anticonvulsants.
Yes. Both modes are suitable, provided temperature and moisture protection is ensured.
It is often used when first-line NNRTI/NRTI regimens fail or resistance is observed, making it crucial in salvage therapies.
Because we offer: WHO-GMP compliant manufacturing; Strict impurity and stability control; Competitive pricing; Full regulatory documentation; Reliable global logistical support; Ability to supply both Free Base & Mesylate forms
Looking to source Saquinavir or other high-quality pharmaceutical products?
We’re here to help.
Whether you need Active Pharmaceutical Ingredients (APIs), finished formulations, or regulatory support,
our team is ready to provide dependable, compliant, and cost-effective solutions tailored to your market needs.
The contents of this page and any attachments are intended solely for the designated recipient(s) and may contain confidential and/or privileged information protected by law.
Any patented products are excluded from our offerings in regions where such patents are currently in force.