
API's
Active Pharmaceutical Ingredients

Active Pharmaceutical Ingredients
API
USP / EP / BP
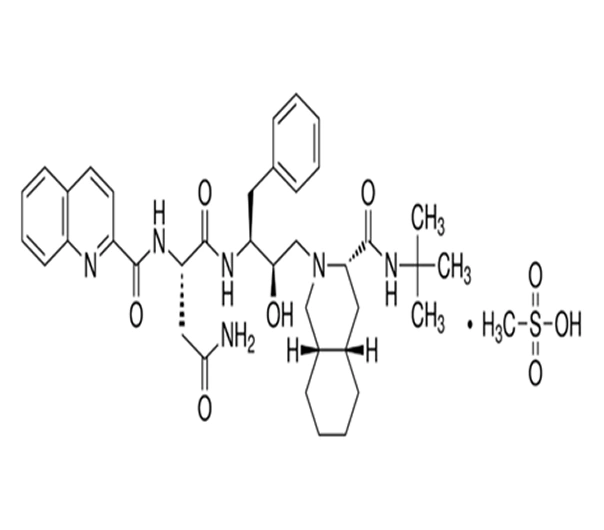
C38H50N6O5·CH4O3S
(2S)-N-[(2S,3R)-4-[(3S)-3-[(2S)-2-[(methoxycarbonyl)amino]-3-methylbutanoyl]amino]-3-hydroxy-1-phenylbutan-2-yl]-2-[[(2S)-3-methyl-2-[[(phenylmethyl)oxy]carbonyl]amino]butanoyl]amino]-4-(quinolin-2-ylmethyl)butanamide methanesulfonate
149845-06-7
767.96 g/mol
HIV-1 Protease Inhibitor (Antiviral)
| Appearance | White to off-white crystalline powder |
|---|---|
| Solubility | Freely soluble in methanol & ethanol; slightly soluble in water |
| Melting Point | 195–200°C |
| pH | 5.0–6.5 |
Saquinavir Mesylate is a potent HIV-1 protease inhibitor used in antiretroviral therapy (ART). It works by blocking the viral protease enzyme required for HIV polyprotein cleavage, preventing the virus from maturing into an infectious form. Salius Pharma supplies high-purity Saquinavir Mesylate API as a white to off-white crystalline powder, compliant with USP/EP/IP pharmacopeial standards.
Reduces viral load; Enhances immune recovery (increased CD4 count); Works synergistically with ritonavir (“boosted” regimens); Improves long-term HIV management
| Attribute | Why Salius Pharma Is a Good Choice |
|---|---|
| Quality Certification | WHO GMP, ISO 9001:2015, FDA audited facilities |
| Affordability & Generic Access | Cost competitive generics for global supply |
| Export Experience | Proven track record of supplying globally |
When you order from Salius Pharma, you receive more than just a product — you receive a complete, professionally prepared package that meets international regulatory and quality expectations.
We ensure prompt and reliable shipping worldwide. Delivery times vary depending on destination, order volume, and regulatory requirements.
7 to 21 business days from dispatch, depending on your location.
Treatment of HIV-1 infection as part of combination antiretroviral therapy.
Typically 36 months, depending on storage conditions and packaging.
Yes, it is frequently combined with ritonavir (boosted therapy) to enhance bioavailability.
Salius Pharma exports globally, including regulated markets in the US, EU, and Asia.
Yes, client-specific analytical methods or additional test parameters can be incorporated.
Contact Salius Pharma via email or phone with product name, quantity, and delivery details; a quotation and documentation package will be provided.
Looking to source Saquinavir Mesylate or other high-quality pharmaceutical products?
We’re here to help.
Whether you need Active Pharmaceutical Ingredients (APIs), finished formulations, or regulatory support,
our team is ready to provide dependable, compliant, and cost-effective solutions tailored to your market needs.
The contents of this page and any attachments are intended solely for the designated recipient(s) and may contain confidential and/or privileged information protected by law.
Any patented products are excluded from our offerings in regions where such patents are currently in force.