
API's
Active Pharmaceutical Ingredients

Active Pharmaceutical Ingredients
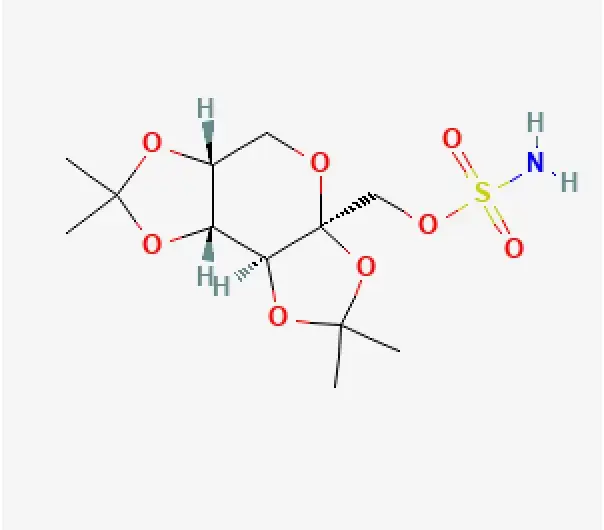
API
Pharmaceutical Grade
C12H21NO8S
2,3:4,5-Bis-O-(1-methylethylidene)-β-D-fructopyranose sulfamate
97240-79-4
339.36 g/mol
Fructopyranose sulfamate derivative
Antiepileptic / Migraine prophylactic agent
| Appearance | White to off-white crystalline powder |
|---|---|
| Solubility | Slightly soluble in water; freely soluble in acetone, ethanol, and methanol; insoluble in ether |
| Melting Point | 178 – 182°C (with decomposition) |
| pH (1% aqueous solution) | 4.0 – 5.0 |
Topiramate is a sulfamate-substituted monosaccharide derivative with broad-spectrum anticonvulsant and neuromodulatory properties. It is primarily indicated for the treatment of epilepsy and migraine prevention, and is also used in psychiatric and metabolic disorders. Topiramate’s unique mechanism combines voltage-gated sodium channel inhibition, enhancement of GABAergic activity, and inhibition of excitatory glutamate pathways.
Topiramate exhibits a multifactorial mechanism of action that contributes to its antiepileptic and neuroprotective efficacy: (1) blocks voltage-gated sodium channels to stabilize neuronal membranes, (2) enhances inhibitory GABAergic neurotransmission, (3) antagonizes excitatory glutamate (AMPA/kainate) receptors, and (4) weakly inhibits carbonic anhydrase enzymes—collectively reducing neuronal hyperexcitability and seizure propagation.
| Attribute | Why Salius Pharma Is a Good Choice |
|---|---|
| Quality Certification | WHO GMP, ISO 9001:2015, FDA audited facilities |
| Affordability & Generic Access | Cost competitive generics for global supply |
| Export Experience | Proven track record of supplying globally |
When you order from Salius Pharma, you receive more than just a product — you receive a complete, professionally prepared package that meets international regulatory and quality expectations.
We ensure prompt and reliable shipping worldwide. Delivery times vary depending on destination, order volume, and regulatory requirements.
7 to 21 business days from dispatch, depending on your location.
Topiramate is manufactured in WHO-GMP and FDA-audited facilities in India, ensuring global compliance and consistent batch reproducibility.
Pharmaceutical Grade conforming to USP / BP / IP specifications, with analytical methods validated as per ICH guidelines.
Topiramate is non-hazardous and suitable for air or sea shipment under controlled temperature and humidity to maintain product integrity.
10–15 working days depending on stock, documentation, and shipping arrangements.
Yes. We assist with dossier preparation, CTD/ACTD formatting, and country-specific data customization for faster product registration.
Yes. Fully compliant with ICH Q7, EU-GMP, and USFDA guidelines; supported by validated analytical data and stability studies.
We offer ongoing documentation updates, COA revalidation, technical clarifications, and assistance during regulatory audits.
Looking to source Topiramate or other high-quality pharmaceutical products?
We’re here to help.
Whether you need Active Pharmaceutical Ingredients (APIs), finished formulations, or regulatory support,
our team is ready to provide dependable, compliant, and cost-effective solutions tailored to your market needs.
The contents of this page and any attachments are intended solely for the designated recipient(s) and may contain confidential and/or privileged information protected by law.
Any patented products are excluded from our offerings in regions where such patents are currently in force.