
API's
Active Pharmaceutical Ingredients

Active Pharmaceutical Ingredients
API
USP / EP / Veterinary Grade
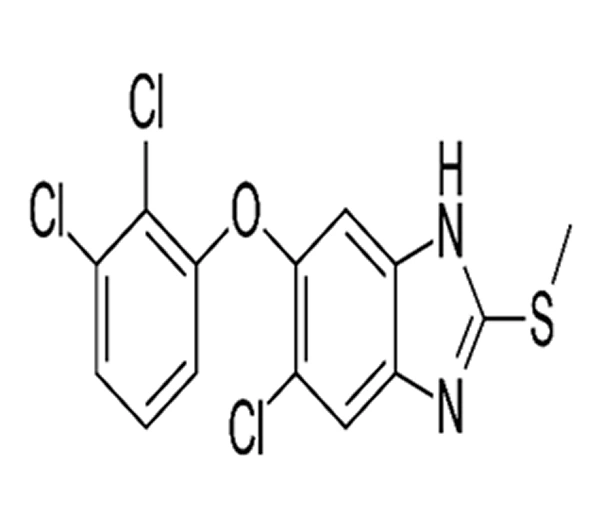
C₁₄H₉Cl₃N₂OS
5-Chloro-2-(2,3-dichlorophenoxy)-6-(methylthio)-1H-benzimidazole
68786-66-3
359.66 g/mol
Antiparasitic
| Appearance | White to off-white crystalline powder |
|---|---|
| Solubility | Practically insoluble in water; Soluble in DMSO and organic solvents |
| Melting Point | 177–183°C |
| pH | Neutral to slightly acidic |
Triclabendazole is a broad-spectrum anthelmintic API widely used for the treatment of fascioliasis (liver fluke infections) in humans and animals. It is highly effective against both immature and adult stages of Fasciola hepatica and Fasciola gigantica, making it one of the most potent flukicides on the market.
Triclabendazole disrupts microtubule-based processes in flukes by: Binding to β-tubulin; Inhibiting microtubule formation; Disrupting key metabolic pathways; Leading to fluke paralysis and death. It is effective at very low doses and works against parasites resistant to other anthelmintics.
| Attribute | Why Salius Pharma Is a Good Choice |
|---|---|
| Quality Certification | WHO GMP compliant, ISO 9001:2015 certified, with FDA audited facilities. |
| Affordability & Generic Access | Cost competitive generics for global supply. |
| Export Experience | Proven track record of supplying globally. |
When you order from Salius Pharma, you receive more than just a product — you receive a complete, professionally prepared package that meets international regulatory and quality expectations.
We ensure prompt and reliable shipping worldwide. Delivery times vary depending on destination, order volume, and regulatory requirements.
7 to 21 business days from dispatch, depending on your location.
It is used to treat liver fluke infections in humans and animals, effective against both mature and immature stages.
It disrupts parasite microtubule formation and energy metabolism, leading to immobilization and death of liver flukes.
Pharmaceutical grade, EP/USP compliant, and veterinary grade.
Yes. We export to Asia, Africa, the Middle East, Europe, North America, South America, and other regions with suitable regulatory and logistics support.
Yes, micronized grades and customized mesh sizes are available upon request.
COA, MSDS, TDS, stability data, origin certificate, and all standard export and shipping documents.
Looking to source Triclabendazole or other high-quality pharmaceutical products?
We’re here to help.
Whether you need Active Pharmaceutical Ingredients (APIs), finished formulations, or regulatory support,
our team is ready to provide dependable, compliant, and cost-effective solutions tailored to your market needs.
The contents of this page and any attachments are intended solely for the designated recipient(s) and may contain confidential and/or privileged information protected by law.
Any patented products are excluded from our offerings in regions where such patents are currently in force.